Options, Choices and Treatments for Cancer Recovery: De mystifying the oscillating the immune cycle.
When my ex husband and I had the idea for starting support groups for cancer patients in the early 1980’s. cancer organizations, patients and doctors were initially not supportive. In fact they were skeptical. They could not see any possible therapeutic benefit could come from people attending a support group. How wrong they were! Move forward 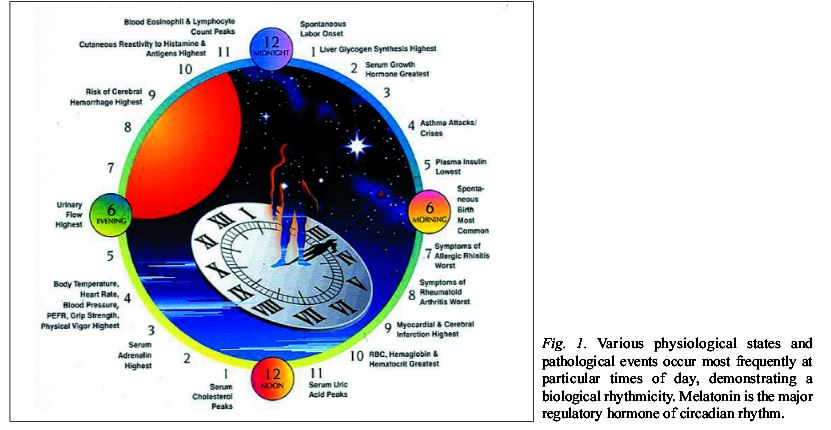 40 years! Yes this December marks my beginning working with cancer patients 40 years ago in a time when there was no support in the health system and no support groups. Now, the benefit of patients attending structured supports and the well being benefit is indisputable.
40 years! Yes this December marks my beginning working with cancer patients 40 years ago in a time when there was no support in the health system and no support groups. Now, the benefit of patients attending structured supports and the well being benefit is indisputable.
There are still many new areas to explore, which leads me to discussing the immune cycle. It would seem that during 2014 on Navigating the Cancer Maze – we uncovered and delivered some significant “missing” pieces of the cancer treatment puzzle. It is my hope that as 2014 comes to an end – that 2015 will truly usher in a new paradigm in cancer treatments. The foundation has been laid, extensive research already done, the concept has been introduced worldwide – the immune cycle measurement is here – NOW!
Now it is up to cancer patients to prove Martin Ashdown and Brendon Coventry right….or wrong. I liked it when Martin Ashdown said – “We believe this is so, a breakthrough – but we are open to being proven wrong!” As in the early days of my first charitable foundation – it was people power – patient power that made a difference to the success of the Cancer Support Group Movement. Once told there would likely be a measured benefit from attending a support group – the press broadcast the news and the patients came on board en masse!
New cancer treatments are always controversial; BUT – this method is not a treatment. It’s a smart approach. A Smart approach that utilizes all that we know so far about cancer. It works because ultimately; our innate immune system knows what to do. It is just in temporary overwhelm. So doesn’t it make perfect sense that to find the best time to work in synch within the cycle of each person’s immune system to add the best chemotherapy or monoclonal antibody or other targeted treatment. Isn’t this the personalized approach we have been searching for? We all thought it would come in a pill – not an approach and that’s a paradigm shift that some in the world of science and medicine are struggling with.
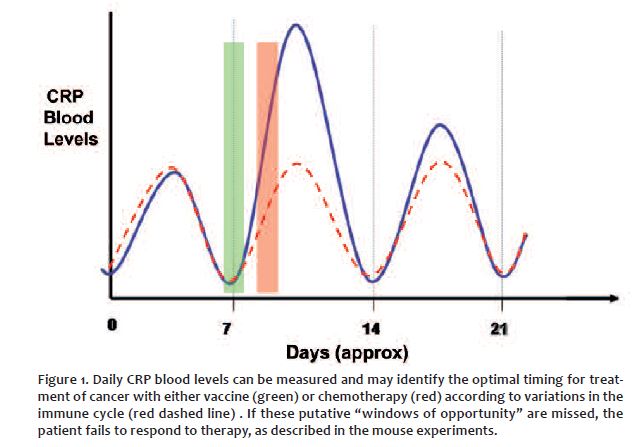 Knowing how one’s immune cycle oscillates holds a clue, “the missing link” that can direct doctors to seek the best time to treat patients in their personal window of opportunity. Then within the parameters of what we know they will have the best chance of a good response or complete response to treatment. (CR)
Knowing how one’s immune cycle oscillates holds a clue, “the missing link” that can direct doctors to seek the best time to treat patients in their personal window of opportunity. Then within the parameters of what we know they will have the best chance of a good response or complete response to treatment. (CR)
When cancer cells challenge us – they are also smart. They are a part of us created by our internal systems. You could define cancer as an internal systems error! These cancer cells cleverly recruit our intelligent mechanisms using them for their own growth and survival. It seems a bit crazy that something that wants to survive – kills its host. But – that’s life! To listen to the latest Voice America related to this blog visit:
http://www.voiceamerica.com/episode/82056/options-choices-and-treatments-for-cancer-recovery
So – I believe as do Ashdown and Coventry – that if there is enough of the immune response left in a patient – that the cycle can still be measured and timed so administer the right treatment at the right time. It is simple enough – the only real cost being a series of blood tests. Then finding a doctor who will look at the science and research and say yes – they administer treatment during the 12 hour window of opportunity. The Grace Gawler Institute is keen to let you know and experience the immune cycle for yourself. Please Read more below or join the immune cycle registry at:
http://www.gracegawlerinstitute.com/immune-cycle-registry/ also see our Next “Survivor Academy” Course!
Martin Ashdown and Brendon Coventry have built upon earlier excellent work in the study of chronobiology: Below are references given on Navigating the Cancer Maze today: I have provided abstract content as well as links.
Until next time……Grace
- Annu Rev Pharmacol Toxicol. 2010;50:377-421. doi: 10.1146/annurev.pharmtox.48.113006.094626.
Circadian timing in cancer treatments.
Lévi F1, Okyar A, Dulong S, Innominato PF, Clairambault J.
Author information
Abstract
The circadian timing system is composed of molecular clocks, which drive 24-h changes in xenobiotic metabolism and detoxification, cell cycle events, DNA repair, apoptosis, and angiogenesis. The cellular circadian clocks are coordinated by endogenous physiological rhythms, so that they tick in synchrony in the host tissues that can be damaged by anticancer agents. As a result, circadian timing can modify 2- to 10-fold the tolerability of anticancer medications in experimental models and in cancer patients. Improved efficacy is also seen when drugs are given near their respective times of best tolerability, due to (a) inherently poor circadian entrainment of tumors and (b) persistent circadian entrainment of healthy tissues. Conversely, host clocks are disrupted whenever anticancer drugs are administered at their most toxic time. On the other hand, circadian disruption accelerates experimental and clinical cancer processes. Gender, circadian physiology, clock genes, and cell cycle critically affect outcome on cancer chronotherapeutics. Mathematical and systems biology approaches currently develop and integrate theoretical, experimental, and technological tools in order to further optimize and personalize the circadian administration of cancer treatments.
PMID:
20055686
[PubMed – indexed for MEDLINE] - http://www.ncbi.nlm.nih.gov/pubmed/20055686
Ann Pharm Fr. 2008 Jun;66(3):175-84. doi: 10.1016/j.pharma.2008.05.003.
[The circadian-timing system: a determinant of drug activity and a target of anticancer treatments].
[Article in French]
Lévi F.
Author information
Abstract
Cellular proliferation and drug detoxification are controlled over the 24h by the circadian-timing system, whose disruption can favor malignant processes. Thus, prolonged shift work appears to increase the risk of breast, colon or prostate cancer. Alterations in circadian physiology and/or molecular-clock genes accelerate cancer progression in experimental models and in cancer patients. In addition, anticancer treatments can also dampen or reinforce the circadian-timing system, as a function of dose and time of administration. The adjustment of anticancer-drug delivery to the circadian-timing system (chronotherapeutics) has allowed to reduce five-fold the incidence of severe adverse events as compared to constant rate infusion or wrongly-timed chronomodulated delivery in cancer patients. In experimental models, the best antitumor efficacy is usually obtained following treatment delivery near the least toxic time, a statement that also seems to apply to patients. Dedicated technologies include programmable in time pumps and rhythm monitors and are required for chronotherapeutics. Recent results have revealed that the optimal chronotherapeutic schedule could differ as a function of gender and circadian physiology. In conclusion, the circadian-timing system was shown to negatively control malignant proliferation via partly identified molecular mechanisms. The components of the circadian-timing system thus constitute new potential therapeutic targets in oncology. Mathematical models help toward a better understanding of the role of variability for the determination of the optimal chronotherapeutic schedule and constitute useful tools for the personalization of cancer chronotherapeutics.
http://www.ncbi.nlm.nih.gov/pubmed/18706346
Handb Exp Pharmacol. 2013;(217):261-88. doi: 10.1007/978-3-642-25950-0_11.
Cancer chronotherapeutics: experimental, theoretical, and clinical aspects.
Ortiz-Tudela E1, Mteyrek A, Ballesta A, Innominato PF, Lévi F.
Author information
Abstract
The circadian timing system controls cell cycle, apoptosis, drug bioactivation, and transport and detoxification mechanisms in healthy tissues. As a consequence, the tolerability of cancer chemotherapy varies up to several folds as a function of circadian timing of drug administration in experimental models. Best antitumor efficacy of single-agent or combination chemotherapy usually corresponds to the delivery of anticancer drugs near their respective times of best tolerability. Mathematical models reveal that such coincidence between chronotolerance and chronoefficacy is best explained by differences in the circadian and cell cycle dynamics of host and cancer cells, especially with regard circadian entrainment and cell cycle variability. In the clinic, a large improvement in tolerability was shown in international randomized trials where cancer patients received the same sinusoidal chronotherapy schedule over 24h as compared to constant-rate infusion or wrongly timed chronotherapy. However, sex, genetic background, and lifestyle were found to influence optimal chronotherapy scheduling. These findings support systems biology approaches to cancer chronotherapeutics. They involve the systematic experimental mapping and modeling of chronopharmacology pathways in synchronized cell cultures and their adjustment to mouse models of both sexes and distinct genetic background, as recently shown for irinotecan. Model-based personalized circadian drug delivery aims at jointly improving tolerability and efficacy of anticancer drugs based on the circadian timing system of individual patients, using dedicated circadian biomarker and drug delivery technologies.
http://www.ncbi.nlm.nih.gov/pubmed/23604483
Chronobiol Int. 2002 Jan;19(1):1-19.
From circadian rhythms to cancer chronotherapeutics.
Lévi F.
Author information
Abstract
Mammalian circadian rhythms result from a complex organization involving molecular clocks within nearly all “normal” cells and a dedicated neuroanatomical system, which coordinates the so-called “peripheral oscillators.” The core of the central clock system is constituted by the suprachiasmatic nuclei that are located on the floor of the hypothalamus. Our understanding of the mechanisms of circadian rhythm generation and coordination processes has grown rapidly over the past few years. In parallel, we have learnt how to use the predictable changes in cellular metabolism or proliferation along the 24h time scale in order to improve treatment outcome for a variety of diseases, including cancer. The chronotherapeutics of malignant diseases has emerged as a result of a consistent development ranging from experimental, clinical, and technological prerequisites to multicenter clinical trials of chronomodulated delivery schedules. Indeed large dosing-time dependencies characterize the tolerability of anticancer agents in mice or rats, a better efficacy usually results from treatment administration near the least toxic circadian time in rodent tumor models. Programmable in time multichannel pumps have allowed to test the chronotherapy concepts in cancer patients and to implement chronomodulated delivery schedules in current practice. Clinical phase I and II trials have established the feasibility, the safety, and the activity of the chronotherapy schedules, so that this treatment method has undergone further evaluation in international multicenter phase III trials. Overall, more than 2,000 patients with metastatic disease have been registered in chronotherapy trials. Improved tolerability and/or better antitumor activity have been demonstrated in randomized multicenter studies involving large patient cohorts. The relation between circadian rhythmicity and quality of life and even survival has also been a puzzling finding over the recent years. An essential step toward further developments of circadian-timed therapy has been the recent constitution of a Chronotherapy cooperative group within the European Organization for Research and Treatment of Cancer. This group now involves over 40 institutions in 12 countries. It is conducting currently six trials and preparing four new studies. The 19 contributions in this special issue reflect the current status and perspectives of the several components of cancer chronotherapeutics.
PMID:
11962669
[PubMed – indexed for MEDLINE]
http://www.ncbi.nlm.nih.gov/pubmed/11962669
Cancer Causes Control. 2006 May;17(4):611-21.
Chronotherapeutics: the relevance of timing in cancer therapy.
Lévi F.
Author information
Abstract
BACKGROUND:
Cell physiology is regulated along the 24-h time scale by a circadian timing system composed of molecular clocks within each cell and a central coordination system in the brain. The mammalian molecular clock is made of interconnected molecular loops involving at least 12 circadian genes. The cellular clocks are coordinated by the suprachiasmatic nuclei, a hypothalamic pacemaker which also helps the organism adjust to environmental cycles. The rest-activity rhythm is a reliable marker of the circadian system function in both rodents and man. This circadian organization is responsible for predictable changes in the tolerability and efficacy of anticancer agents, and possibly also in tumor promotion or growth.
METHODS:
Expected least toxic times of chemotherapy were extrapolated from experimental models to human subjects with reference to the rest-activity cycle. The clinical relevance of the chronotherapy principle, i.e. treatment administration as a function of rhythms, has been demonstrated in randomized multicenter trials.
RESULTS:
Chronotherapeutic schedules have been used to safely document the activity of the association of oxaliplatin, 5-FU and leucovorin against metastatic colorectal cancer and to set up a new medicosurgical management of this disease which achieved unprecedented long term survival.
CONCLUSION:
The chronotherapy concept offers further promises for improving current cancer treatment options as well as for optimizing the development of new anticancer or supportive agents.
PMID:
16596317
[PubMed – indexed for MEDLINE]
Mol Med. 2012 Dec 6;18:1249-60. doi: 10.2119/molmed.2012.00077.
Circadian rhythm disruption in cancer biology.
Savvidis C1, Koutsilieris M.
Author information
Abstract
Circadian rhythms show universally a 24-h oscillation pattern in metabolic, physiological and behavioral functions of almost all species. This pattern is due to a fundamental adaptation to the rotation of Earth around its own axis. Molecular mechanisms of generation of circadian rhythms organize a biochemical network in suprachiasmatic nucleus and peripheral tissues, building cell autonomous clock pacemakers. Rhythmicity is observed in transcriptional expression of a wide range of clock-controlled genes that regulate a variety of normal cell functions, such as cell division and proliferation. Desynchrony of this rhythmicity seems to be implicated in several pathologic conditions, including tumorigenesis and progression of cancer. In 2007, the International Agency for Research on Cancer (IARC) categorized “shiftwork that involves circadian disruption [as] probably carcinogenic to humans” (Group 2A in the IARC classification system of carcinogenic potency of an agent) (Painting, Firefighting, and Shiftwork; IARC; 2007). This review discusses the potential relation between disruptions of normal circadian rhythms with genetic driving machinery of cancer. Elucidation of the role of clockwork disruption, such as exposure to light at night and sleep disruption, in cancer biology could be important in developing new targeted anticancer therapies, optimizing individualized chronotherapy and modifying lighting environment in workplaces or homes.
PMID:
22811066
[PubMed – indexed for MEDLINE]
PMCID:
PMC3521792
Free PMC Article
Chronobiol Int. 2012 Apr;29(3):227-51. doi: 10.3109/07420528.2012.658127.
Clock genes and clock-controlled genes in the regulation of metabolic rhythms.
Mazzoccoli G1, Pazienza V, Vinciguerra M.
Author information
Abstract
Daily rotation of the Earth on its axis and yearly revolution around the Sun impose to living organisms adaptation to nyctohemeral and seasonal periodicity. Terrestrial life forms have developed endogenous molecular circadian clocks to synchronize their behavioral, biological, and metabolic rhythms to environmental cues, with the aim to perform at their best over a 24-h span. The coordinated circadian regulation of sleep/wake, rest/activity, fasting/feeding, and catabolic/anabolic cycles is crucial for optimal health. Circadian rhythms in gene expression synchronize biochemical processes and metabolic fluxes with the external environment, allowing the organism to function effectively in response to predictable physiological challenges. In mammals, this daily timekeeping is driven by the biological clocks of the circadian timing system, composed of master molecular oscillators within the suprachiasmatic nuclei of the hypothalamus, pacing self-sustained and cell-autonomous molecular oscillators in peripheral tissues through neural and humoral signals. Nutritional status is sensed by nuclear receptors and coreceptors, transcriptional regulatory proteins, and protein kinases, which synchronize metabolic gene expression and epigenetic modification, as well as energy production and expenditure, with behavioral and light-dark alternance. Physiological rhythmicity characterizes these biological processes and body functions, and multiple rhythms coexist presenting different phases, which may determine different ways of coordination among the circadian patterns, at both the cellular and whole-body levels. A complete loss of rhythmicity or a change of phase may alter the physiological array of rhythms, with the onset of chronodisruption or internal desynchronization, leading to metabolic derangement and disease, i.e., chronopathology.
PMID:
22390237
[PubMed – indexed for MEDLINE]